"Getting your digital health app prescribed by an HCP is something most digital health app vendors want. Three countries in Europe with more than 149 million public health insured citizens make it possible. See how it works in Germany, Belgium and France.
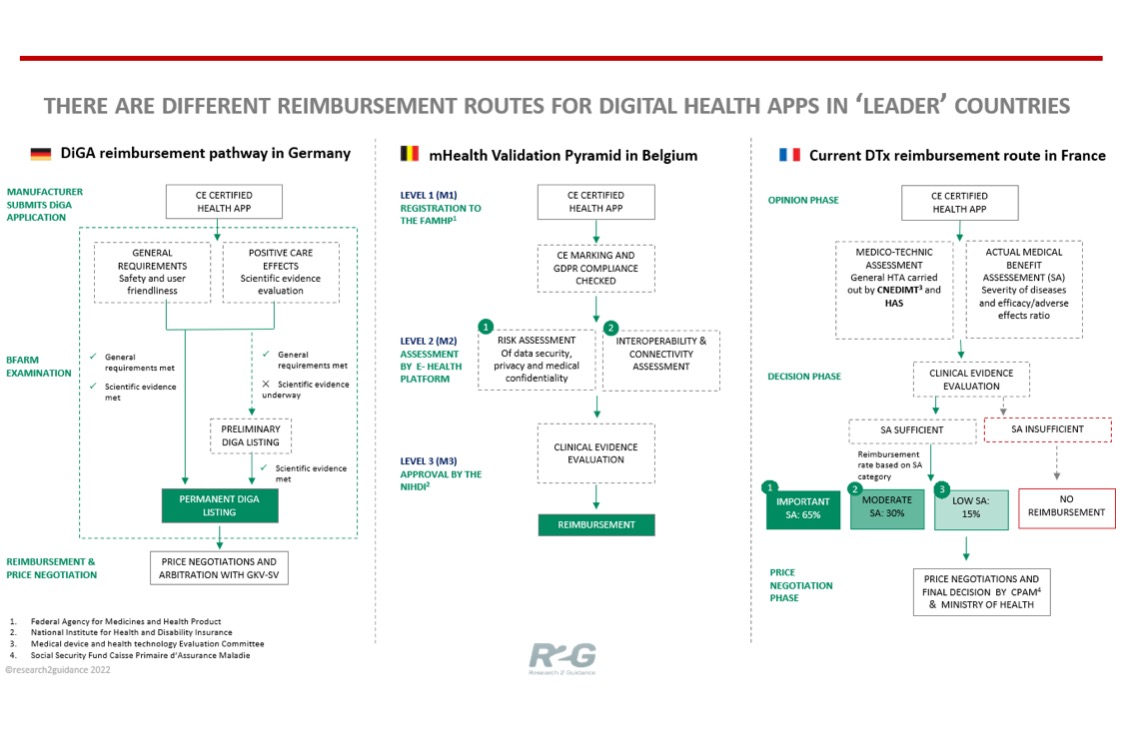
An EU wide initiative to allow HCPs to prescribe digital health apps would brace the reimbursement route as a market entry strategy for many digital health solution providers that seek their DTx solution to get reimbursed by the statutory health insurance. R2G’s “Where is the money in digital health? The roadmap to digital health app reimbursement in Europe.” explains that Germany, Belgium and France are the DTx reimbursement process leaders that have a standardized reimbursement process in place or soon to be launched. Currently, the German DiGA directory consists of 28 apps whereas 34 health apps are present across M1 and M2 levels of mHealth validation pyramid in Belgium. The digital health innovation manufacturers, however, should be aware of the differences among the reimbursement processes in different countries. These differences can be attributed to country-specific regulatory authorities in the EU.
The German DiGA Fast Track process starts with the CE certified app fulfilling two criteria; first, a set of general requirements including data protection, information security, interoperability, and ease of use. Secondly, the solution must prove proof of positive care effects by improving the health of the user. The app which is able to fulfil both criteria is listed in the DiGA directory upon an assessment from BfARM within 3 months. Alternatively, a solution can be preliminarily listed as a DiGA if it only meets the safety and functionality requirements. In this situation, the manufacturer will have a period of 12 months to prove the positive health effects of the solution after which it becomes DiGA listed. Finally, reimbursement price negotiations are carried out by the GKV..."
Lire la suite
How to get your digital health app reimbursed in Europe? Start with Germany, Belgium and France.
RESEARCH2GUIDANCE, 15/02/2022
Partagé par :
Beesens TEAM